Some of the most important types of membrane proteins are as follows:
1. Peripheral (Extrinsic) Proteins 2. Integral (Intrinsic) Proteins 3. Asymmetric Distribution of Membrane Proteins 4. Mobility of Membrane Proteins 5. Enzymatic Properties of Membrane Proteins 6. Isolation and Characterization of Membrane Proteins.
1. Peripheral (Extrinsic) Proteins:
Peripheral or extrinsic membrane proteins are generally loosely attached to the membrane and are more readily removed than are the integral proteins. Peripheral proteins are rich in amino acids with hydrophilic side chains that permit interaction with the surrounding water and with the polar surface of the lipid bilayer. Peripheral proteins on the cell’s exterior membrane surface often contain chains of sugars (i.e., they are glycoproteins).
2. Integral (Intrinsic) Proteins:
Integral or intrinsic membrane proteins contain both hydrophilic and hydrophobic regions. The hydrophilic portions of the protein interact with the polar heads of the lipid molecules at each surface of the bimolecular leaflet.
Portions of integral proteins that project beyond the surface of the lipid bilayer are also rich in hydrophilic amino acids. Amino acids in that part of the protein projecting from the outer membrane surface may be linked to chains of sugars. Parts of the protein that are buried in the hydrophobic portion of the lipid bilayer are rich in amino acids with hydrophobic side chains.
These side chains are believed to form hydrophobic bonds with the hydrocarbon tails of the membrane phospholipids. It is speculated that within the hydrophobic interior of the membrane, the secondary structure of integral proteins is alpha helix and/or beta sheet (Fig. 15-12).
In the alpha helix conformation, amino and carboxyl groups along a stretch of the polypeptide’s backbone form hydrogen bonds with one another; in the beta sheet, hydrogen bonds are formed between amino and carboxyl groups in stretches of polypeptide that lie parallel to one another.
In the absence of such hydrogen bonding, these amino and carboxyl groups would have polar properties, their hydrophilic nature being incompatible with the membrane’s hydrophobic interior. Alpha amino and carboxyl groups that are not “neutralized” by hydrogen bonding would be expected only in those parts of the integral protein that extend into the aqueous milieu on either side of the membrane.
Integral Proteins That Span the Membrane:
M. Bretscher first demonstrated the existence of integral proteins that span the entire membrane. In a series of elegant experiments, Bretscher showed that radioactive ligands specific for membrane proteins of the erythrocyte were bound in smaller quantities to intact cells than to disrupted cells. Disruption of the cells was shown to expose portions of the membrane proteins previously facing the cell interior, thereby allowing additional radioactive ligand to associate with the protein.
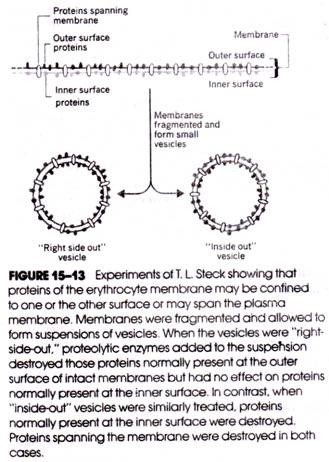
T. L. Steck developed a technique for converting fragments of disrupted erythrocyte membranes into small vesicles that were either “right-side-out” (i.e., the external face of the membrane also formed the external face of the vesicle) or “inside-out” (Fig. 15-13).
When proteolytic enzymes were added to separate suspensions of each type of vesicle, certain of their membrane proteins were found to be equally susceptible to digestion and could therefore be enzymatically attacked from either membrane surface. These proteins clearly spanned the membrane. Other proteins were susceptible to enzymatic digestion only when present in right-side-out or inside-out vesicles, indicating their differential distribution in the membrane’s outer and inner surfaces.
Integral proteins that span the entire membrane contain two outer regions that are hydrophilic (i.e., one at each surface of the membrane); the central region is hydrophobic (Fig. 15-12). Carbohydrate associated with the hydrophilic region facing the cell’s surroundings is believed to play a role in maintaining the orientation of the protein within the membrane. The hydrophilic sugars, together with the hydrophilic side chains of amino acids in the out: region of the protein, effectively prevent reorientation of the protein in the direction of the hydrocarbon core of the lipid bilayer.
3. Asymmetric Distribution of Membrane Proteins:
The outer and inner regions of the plasma membrane do not contain either the same types or equal amounts of the various peripheral and integral proteins. For example, the outer half of the erythrocyte membrane contains far less protein than does the inner, half.
In addition, various membrane proteins may be present in significantly different quantities; the membranes of some cells contain a hundred times as many molecules of one protein species as another. Moreover, regardless of absolute quantity, all copies of a given membrane protein species have exactly the same orientation in the membrane.
The differential distribution of proteins in the various regions of the plasma membrane within a single cell was described earlier in connection with liver parenchymal cells and intestinal epithelium. This irregular distribution of membrane proteins is known as membrane asymmetry. Not only are the proteins of plasma membranes asymmetrically distributed but so too are the proteins of the membranes of the endoplasmic reticulum and vesicular organelles (e.g., mitochondria).
4. Mobility of Membrane Proteins:
When cells are grown in culture, there is an occasional fusion of one cell with another to form a larger cell. The frequency of cell fusion can be greatly increased by adding Sendai virus to the cell culture. In the presence of this virus, even different strains of cells can be induced to fuse, producing hybrid cells or heterokaryons. D. Frye and M. Edidin utilized this phenomenon to demonstrate that membrane proteins may not maintain fixed positions in the membrane but may move about laterally through the bilayer.
Frye and Edidin induced the fusion of human and mouse cells to form heterokaryons and, using fluorescent antibody labels, followed the distribution of human and mouse membrane proteins in the heterokaryon during the time interval that followed fusion.
At the onset of fusion, human and mouse membrane proteins were respectively restricted to their “halves” of the hybrid cell, but in less than an hour both protein types became uniformly distributed through the membrane (Fig. 15-14). The distribution of the membrane proteins was not dependent on the availability of ATP and was not prevented by metabolic inhibitors, indicating that lateral movement of proteins in the membrane occurred by diffusion.
Although some membrane proteins are capable of lateral diffusion, many are not. G. Nicolson and others have obtained evidence suggesting that many integral proteins are restrained within the membrane by a protein network lying just under the membrane’s inner surface (Fig. 15-9). In many cells, this network is associated with a system of cytoplasmic filaments and microtubules that radiate through the cytosol forming a cytoskeleton.
5. Enzymatic Properties of Membrane Proteins:
Membrane proteins have been shown to possess enzymatic activity. Table 15-1 lists some of the enzymes that are now recognized as constituents of the plasma membrane of various cells. To this list of proteins must be added receptor proteins (such as the insulin- binding sites of the liver plasma membrane) and structural or non-enzymatic proteins.
Ectoenzymes and Endoenzymes:
Enzymes disposed in the plasma membrane may be characterized according to the membrane face containing the enzymatic activity. Accordingly, ectoenzymes are those enzymes whose catalytic activity is associated with the exterior surface of the plasma membrane; the activity of plasma membrane endoenzymes is associated with the interior of the cell. Many (perhaps all) plasma membrane ectoenzymes are glycoproteins.
6. Isolation and Characterization of Membrane Proteins:
Because of the relative ease with which they may be purified, the plasma membranes of erythrocytes provided much of the early information on the chemistry of proteins (and lipids) present in membranes. Now, however, plasma membranes can be obtained from many cell types in a reasonably uncontaminated state using various forms of density gradient centrifugation.
Nonetheless, the individual protein constituents of the membrane are not so easily extricated for individual study because of their high degree of insolubility. Varying degrees of success in extracting proteins from the plasma membrane have been achieved using organic detergents (especially sodium dodecyl sulfate, SDS) and concentrated solutions of urea, n-butanol, and ethylene diamine tetraacetic acid (EDTA).
These chemicals have a disaggregating effect on membranes and cause the release of many of the membrane proteins by dissociating the bonds that link the proteins together or to other membrane constituents. Often, the removal of these agents from a preparation of sol- ubilized membrane proteins is quickly followed by the reassociation or reaggregation of the proteins to form an intractable matrix.
Once solubilized, the membrane proteins can be separated into discrete classes using electrophoresis, chromatography, or other procedures. This generally demands that the dissociating agents be present in the separating medium (e.g., the electrophoresis gel, the column eluent, etc.); otherwise, application of the membrane extract to the medium is followed by membrane protein reaggregation into insoluble complexes that will not separate into distinct fractions.
For example, the separation of liver plasma membrane proteins is achieved only if the electrophoresis gel contains SDS. The solubility problem has been one of the greatest barriers to progress in isolating and fully characterizing the proteins of membranes.
Some of the plasma membrane enzymes listed in Table 15-1 have not actually been isolated from the membrane, as removal and isolation of the enzyme is not a prerequisite for establishing its presence. Instead, the enzyme activity can be measured directly in the (un-solubilized) membrane preparation.