In this article we will discuss about the fermentation of olives:- 1. Introduction to Fermentation of Olives 2. Fermentation of Storage and Sicilian-Type Olives (With Process) 3. Fermentation of Spanish-Type Olives (With Process) 4. Control of Spanish-type Fermentations 5. Spoilage Problems of Fermented Olives 6. Softening Spoilage of Fermented Olives 7. Other Abnormalities of Fermented Olives 8. Newer Developments in Fermentation of Olives.
Contents:
- Introduction to Fermentation of Olives
- Fermentation of Storage and Sicilian-Type Olives (With Process)
- Fermentation of Spanish-Type Olives (With Process)
- Control of Spanish-type Fermentations
- Spoilage Problems of Fermented Olives
- Softening Spoilage of Fermented Olives
- Other Abnormalities of Fermented Olives
- Newer Developments in Fermentation of Olives
1. Introduction to Fermentation of Olives:
Although the origin of the art of preparing table olives by lactic acid fermentation is lost in antiquity, the history of the table olive industry in California has been documented. The first olive trees in the state were grown from seed said to have been planted at the Mission San Diego in 1769. The seeds were brought from San Bias, Mexico, by Don Jose de Galvez during an expedition to rediscover the port of Monterey. The seedlings, thus obtained by selection, are the source of the present mission variety of Olea europaea.
Although olives were used for oil production in the California missions as early as 1780, the first production of olive oil outside of the missions did not occur until 1871. The olive was not planted extensively until about 1860, but by 1870 was showing promise of becoming of some importance to California agriculture. Between 1870 and 1900 many olive varieties had been introduced from Europe and Africa and much effort had been made in testing them, principally for oil production, since oil was the major product of the industry.
Pickling of olives for table use, practiced as an art in the missions, on the farms, and in the homes for many years, was of little commercial value until about 1900. (Directions for pickling ripe and green olives are to be found in the early agricultural literature of California.)
For some reason, the art of pickling “ripe” olives was studied more extensively than that of “green” olives. According to Cruess (1958), it was independently discovered about 1900 by Professor Bioletti of the University of California and Mrs. Freda Ehmann, a commercial packer, that ripen olives, after a preliminary treatment with lye (NaOH) to destroy the bitter principle, could be canned and preserved by heat in much the same manner as other foods. This original development, coupled with application of modern science and technology have, somehow, combined to make the California canned ripe olive the major product of the industry.
Formerly, early in this century, oil was still a major product of the olive industry and, until about 1960, still accounted for an appreciable quantity of fruit in each of the yearly product-disposition figures. In years of high yields the olives destined for oil were in an approximately 1 to 1 ratio with olives canned.
However, because of increased harvest costs in California and other competitive factors from abroad, oil production now has become largely a salvage operation, and tons of olives used for oil approximate those used for producing Spanish-type green olives, another salvage product. (Data supporting these statements are available from the Olive Administrative Committee, Fresno, California 93728.)
The disposition of olives for products in approximate order of importance in California comprise black-ripe and green-ripe olives (whole and pitted), Spanish-type, Sicilian-type, Greek-type including brined and salt-cured fruits, and oil.
A portion of the black-ripe olives may be prepared as sliced (cross-section rings), chopped, or segmented (longitudinal) into 4 or more pieces per olive. Canned ripe olives are the major products of the industry and account for 70% or more of all of the olives harvested. All of the green-ripe canned olives are processed and canned at harvest time (called direct or fresh cure by the industry). Fresh cured olives, although not subjected to lactic acid fermentation.
The remainder of the harvested olives, of necessity, has to be stored in salt brine prior to processing. They also undergo lactic acid fermentation. Thus, there are 4 brine fermentations, including “storage,” Sicilian-type, Spanish-type, and Greek-type brined olives.
The “storage” and Sicilian-type fermentations may be considered to be identical for each variety of olive because the fruits are placed directly in brine without lye treatment, whereas the Spanish-type olives are treated with lye to destroy most of the bitterness, washed to remove some of the alkali, and then brined.
The brine-cured Greek-type olives are placed in high salt brine which may not undergo a lactic fermentation, but a fermentation caused by salt tolerant yeasts. Salt-cured Greek-type olives are cured (desiccated) with coarse salt, so they do not undergo fermentation in the strict sense. Therefore, they will not be considered here.
2. F
ermentation of Storage and Sicilian-Type Olives (With Process):
The “storage” and Sicilian-type fermentations are considered identical for each variety of olive because the fruits are placed directly in brine without lye treatment to destroy the bitterness of the olives. If there are no cherry red colored olives in a tank, the disposition of the fruit can be for Sicilian-type olives, or the fruit may be processed into canned black-ripe olives, according to the economic demand.
The open fermentation tanks are filled and headed in much the same manner as already described for cucumbers. The newer fiberglass tanks are filled with olives, brined, and the cover locked in place without the use of a false head to hold the olives submerged in the brine.
The salt concentration of the brine will vary according to the variety of olives and the final disposition to be made of the tank of olives. Sevillano and Ascolano olives are subject to salt shrivel so it is customary to use a lower concentration of salt (4 to 5% NaCl) if either variety is to be sold as Sicilian-type olives.
Otherwise the brine strength will range between 5 and 8% salt because, in the processing of black-ripe olives, salt shrivel is reduced to a minimum by use of a needling machine to puncture the skin of the fruits. This facilitates osmotic exchange between the needled olives and the processing solution, generally water, or, at most, 10° to 12° salometer salt brine. The shrivel is markedly reduced or eliminated by this manipulation.
The olives, whatever their final disposition, undergo a lactic acid fermentation. The amount of total acidity produced is quite variable but will usually range between 0.2 and 0.7% (calculated as grams lactic acid per 100 ml of brine). The development of acidity in (holding) storage brines varies widely because of a number of factors.
The Sevillano variety does not fresh cure well, so the majority of processors hold this variety in salt brine for a minimum of 30 days before putting the fruit through the black-ripe, canned olive process. This holding period, obviously, does not favor acidity in the upper range, for olives ferment slowly at the ambient temperatures prevailing in the brines, especially during the final weeks of the harvest.
If held in open tanks, the acidity may be lost by oxidative yeasts, which decompose the acid developed. This is quite an important factor when open tanks of such olives are held in shaded or covered areas during the colder months when the oxidative yeasts and molds are still more active than the desirable lactic acid bacteria.
The tendency now is to use anaerobic methods to prevent this loss of acidity, either by use of plastic film to cover the surface of the brine or by the use of polyethylene or fiberglass containers designed to minimize the access of air to the fermenting olives.
The changes in the microbiological populations observed in Sicilian-type or holding solution fermentations are quite similar, and the olives are subject to the same microbial spoilage problems. The lactic acid bacteria found in these fermentations are of the same kinds already described for the cabbage (sauerkraut) and cucumber (pickle) fermentations.
Detailed studies made, but never published; indicate that there is a sequence of microorganisms. The initial stage of fermentation is dominated by organisms other than the lactic acid bacteria. In 4 or 5 days, if the brine strength is not more than 5% NaCl, a population of lactic acid bacteria begins to appear.
Leuconostoc mesenteroides, Pediococcus cerevisiae, and Lactobacillus plantarum have been found, but L. mesenteroides never dominates the fermentation, as it does with cabbage during the initial stage of fermentation. P. cerevisiae and L. plantarum, or the latter alone, are always found. The initial population of extraneous organisms, mainly coliform bacteria and bacilli, disappear gradually and, if the fermentation is normal, no longer can be found after 10 to 14 days of fermentation.
Fermenting yeasts appear in the first 1 to 2 weeks and continue throughout the fermentation. Some of these yeasts are acid-formers, but their contribution to the increase in total acidity of the olive fermentations may be offset by the decomposition of the acidity by oxidative yeasts, which also may become established if conditions are favorable for their growth.
The total acidity developed during the fermentation of holding solution olives destined for future processing varies according to the time the fruit is held in brine. The total acidity may be about 0.1% (total acidity calculated as lactic, g/100 ml) when held for minimum of 30 days as is required for the Sevillano variety or, if the olives are held longer, and the oxidative yeasts and molds are controlled, the total acidity may be as high as 0.6% as lactic acid.
3. Fermentation of Spanish-Type Olives
(With Process):
The first extensive commercial-scale experiments made to study this fermentation were reported by Cruess (1930). From about 1935 until 1955 there was a gradual increase in the quantity of Spanish-type olives fermented in California. All sizes of fruit of the Manzanilla, Sevillano, and Barouni varieties were fermented as Spanish-type green olives, the Barouni variety in particular.
However, rising production costs, and the adoption of the industry-wide marketing agreement limiting the sizes of each variety that may be canned in California made it unprofitable to continue making Spanish-type olives from all sizes except for the Barouni olives; which were exempt because of their unsuitability for canning. At present, the production of Spanish-type olives is strictly a salvage operation, and the non-canning sizes of all varieties are utilized.
Also, for economic reasons, the use of the traditional 190 liter (50 gal.) barrels for fermentation has been discontinued. The development of plastics fostered the replacement of the wood barrel with rigid plastic, bottle-shaped containers of 1514 liter (400 gal.) capacity made of polyethylene, with polyvinyl chloride fittings for draining. These bottles have the advantage of holding the equivalent of 8 barrels of olives, being mobile with the aid of lift trucks, even when filled with olives and brine.
Another advantage is that use of the bottles permits lye treatment, washing (leaching), and brining in the same container as was done earlier by using large redwood tanks holding the equivalent of about 55 barrels of olives. Also there is a considerable economy realized because the bottles require no maintenance upkeep when not in use.
The bottles are filled with olives of the appropriate size, transported to the lye treatment and washing area, and the olives are covered with a lye solution. The concentration of lye used for treating green olives to hydrolyze the bitter glucoside, oleuropein, varies between 0.9 and 1.25% as used in California. Stronger lye solutions must be used very carefully, for they frequently cause softening and blistering, as well as undesirable skin sloughing with all varieties.
Fruit treated during the early part of the season (September 15 to October 15) is prone to blistering when a gaseous fermentation starts because the skin has separated from the flesh due to lye temperature and, if the skin remains intact, gas formation causes a blister to form. This form of blister formation, commonly known as “fish-eye” spoilage.
Cooling of the lye solution and/or olives is indicated. The practice in the San Joaquin Valley has been to use block ice or a heat-exchanger to cool the lye solutions during the early part of the season when the ambient temperatures still remain high during the day.
The lye is allowed to penetrate about 1/2 to 3/4 of the way to the pits of all varieties treated with the exception of the Barouni. This variety must be treated to the pit, for the flesh, unless so treated, will become an undesirable reddish purple color in the area not exposed to lye and the color will intensify as the acidity of the brine increases. This color change is thought to be caused by a leuco-anthocyanin present in the flesh of the fruit.
The time required for the desired lye penetration varies according to the concentration and temperature of the lye solution and the variety, size, maturity, and temperature of the fruit. However, an attempt is made to maintain a schedule to complete the penetration in 12 to 14 hr.
Formerly, the removal of residual lye by washing and leaching with water was carried out by changing the leaching water every 3 to 6 hr during the day. The interval at night might reach 10 hr between changes of water. The washing-leaching extended for 24 to 48 hr before the olives were brined.
By 1943, the trend was to shorten the washing- leaching period in order to minimize the graying of the color of the fruit. Now, because the number of changes of leaching water has been reduced to 3 or 4 changes in 24 hr, the olives are quite alkaline when brined.
Formerly, the industry used 2 different concentrations of salt when brining olives for the Spanish-type fermentation. The Sevillano variety is susceptible to salt shrivel in brine having more than 5% NaCl. Therefore, the processors started this variety in a low concentration of salt (4.0 to 5%), and then increased the salt in the brine slowly to 7-8% over an indefinite interval.
At present, the majority of all varieties used in the industry are brined with about 10% salt solution which is acidified with enough lactic acid to neutralize the residual lye remaining after the washing-leaching period. Most processors add sufficient acid to the brine to lower the pH of the stabilized olives and brine to a value of between 4.5 and 5.0.
Sometimes glucose (crystalline corn sugar) is added at the time of brining. If this is done, the sugar is added at the rate of 0.45 kg (1 lb) (approximately) per 189.3 liters (50 gal.) of olives and brine. However, the limitation of loss of fermentable materials by use of a much shorter, less rigorous washing and leaching before brining has largely eliminated the need for addition of sugar during the fermentation, especially when the plastic bottle is used.
In normal fermentation of green olives of the Spanish-type, the initial stage of the pickling process is the most important phase. During this stage, lasting up to 14 days, if the brines are not acidified, the original contaminating population of Gram-negative and Gram-positive bacteria is eliminated as the result of acid production, both by themselves and by the developing population of lactic acid bacteria.
Formerly, Leuconostoc mesenteroides and Streptococcus faecalis were always found in low salt fermentations of the Sevillano variety. L. mesenteroides, which dominated the latter phase of the primary stage, as well as the early part of the secondary stage of fermentation, disappeared from the population within 3 or 4 weeks.
Lactobacillus plantarum, a non-gas- forming species, dominated the latter part of the intermediate stage as well as the final stage of fermentation. This species is known to persist in olive brines for more than a year. L. brevis, a gas-forming type, is found in the latter phases of the intermediate stage and is present in appreciable numbers during the final stage of fermentation.
This latter species never approaches the population levels produced by L. plantarum. Furthermore, as shown by Vaughn et al. (1943), L. brevis was never found in the high salt fermentations conducted with the Manzanilla variety. Table 6.5 summarizes the predominating bacterial population trends described, and also shows the accompanying changes in the acidity and pH values.
Pediococci were not found in any of the fermentations described by Vaughn et al. (1943). However, in later studies it was found that Pediococcus cerevisiae may be isolated from olive brines during the last phases of the initial stage of fermentation and the first part of the intermediate stage of fermentation, and then it declines rapidly.
Not all brines of Spanish-type olives contain P. cerevisiae. Also, since all varieties of olives are started in 10% salt brines now, the gas- forming species L. brevis also may not be found in the fermentations. Now, the only lactic acid bacterium sure to be found in all fermentations is L. plantarum.
4. Control of Spanish-type Fermentations:
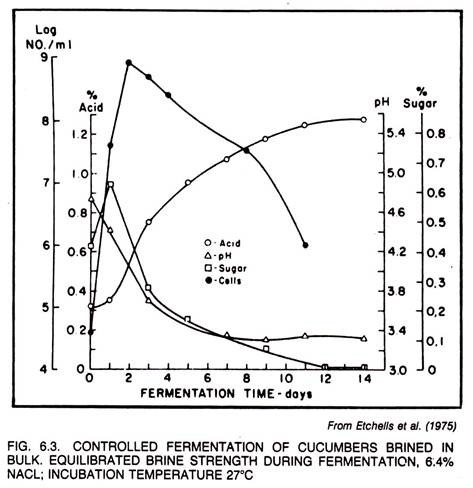
The olive fermentation is very slow in comparison to either the sauerkraut or pickle fermentation. Data in the literature indicate that either the sauerkraut or pickle fermentation will have produced maximum total acidity before the olive fermentation has passed through the initial stage of fermentation.
Olives brined for Spanish-type fermentations in late September and early October produce more acid because of more favorable ambient temperatures. The olives brined from the middle of October until the end of the harvest ferment more slowly and may become dormant soon after the start of fermentation because of the approach of winter and the resultant drop in temperature. Therefore, it is obvious that temperature control is necessary to prevent dormancy of the fermentations.
The fermentations of all varieties of Spanish-type olives brined in California may be accelerated by proper incubation. Commercial scale experiments made by Cruess (1930) indicated that an average temperature range of 21° to 24°C (70° to 75°F) was satisfactory for acceleration of acid production without impairing the quality of the fermented olives. Additional studies by Vaughn et al. (1943) showed that the optimum temperature for maximum acid production of pure cultures of Leuconostoc mesenteroides and Lactobacillus plantarum was 30°C (86°F), and for L. brevis was 34°C (93.2°F).
Temperatures much above 32.2°C (90°F) have been observed to be undesirable for pickling. Therefore, the temperature recommended by Cruess can be followed with assurance. A temperature range of 23.9° to 30°C (75° to 86°F) is commonly used for incubation at present, but only to the extent that fermented Spanish-type olives are required for early delivery.
Fermenting olives should not be incubated until the potential spoilage bacteria, which probably are always present at the beginning of the fermentation, have been eliminated. With normal uncontrolled fermentations, the time required for disappearance of the undesirable bacteria varies from 1 to as much as 3 weeks, depending upon the activity of the lactic acid bacteria and the availability of fermentable material. Those processors who acidify the initial brine may, if desirable, initiate incubation at the start of the fermentation.
Fermentation of Spanish-type olives may also be accelerated by use of starter cultures and addition of fermentable sugar. Cruess (1930) recommended the use of starters of normal brine. Later (1937), Cruess suggested the use of pure cultures of lactic acid bacteria for ensuring start of fermentation. Pure culture starter inoculations were used extensively in California from 1937 until about 1955, particularly with the Manzanilla variety.
Lactobacillus plantarum was the species of choice to use for starters. Details of preparation and use of the starters in the industry have been described by Vaughn et al. (1943). Now, although sophisticated pure culture starters are available for commercial culture laboratories, for economic reasons, when inoculation is indicated, normal brine is used to reseed the suspect fermentation.
Cruess (1930) was the first to recognize the need for addition of supplementary fermentable sugar to obtain satisfactory acid formation in Manzanilla olives and in Sevillano brines which did not otherwise develop enough acidity. Supplementary glucose (from corn) is used extensively, especially with the Barouni, Manzanilla, and Mission varieties.
The sugar is commonly added at the rate of 0.45 to 0.9 kg (1 to 2 lb) for each 189.3 liters (50 gal.) of olives and brine. Unless acidification of the initial brine is practiced, the processor waits from 1 to 3 weeks before adding the sugar. For the best olives and avoidance of malodorous spoilage, the fermentations are controlled to a pH of at least 4.0 and, preferably, 3.8.
Acidification of the brine was first suggested by Cruess (1930) as means of preventing potential loss of fruit caused by abnormal fermentation. It already had been reported by Fornachon et al. (1940) that Lactobacillus brevis and other gas-forming lactobacilli had an optimum pH in the range of 5.0 to 6.0 for growth and decomposition of different carbon compounds.
This latter study was considered to be good evidence for the possible need to acidify all olive brines. Therefore, additional studies, for the most part under commercial conditions, were made by Vaughn et al. (1943). They found that when acidification of the brine, with either acetic or lactic acid, was accompanied by use of a starter culture of L. plantarum and addition of supplementary glucose, satisfactory fermentations were obtained. Acidification apparently functions to spark the activity of the lactic acid bacteria as well as to eliminate undesirable spoilage organisms.
Perhaps the most important control of all for protecting olives in fermentation storage, Sicilian- or Spanish-type, is the maintenance of anaerobic or nearly anaerobic conditions. Control of oxidative molds and yeasts is mandatory but only recently became recognized as such by the industry as a whole.
The advent of plastic film and molded plastic containers during the 1960s was responsible for important changes in the fermentation of green olives in California The use of pliable plastic film to cover the brines in the open bulk fermentation tanks (redwood) for the first time permitted an airtight closure that, once in place, needed little maintenance. This was a major advance because it virtually eliminated oxidative molds and yeasts as sources of potential spoilage. (Plastic films are also used on the open redwood tanks used for storage and Sicilian-type fermentation for the same reason.)
When the industry was using 189.3 liter (50 gal.) barrels exclusively, the first attempt to restrict brine surface exposure to the air was by use of cellar bungs commonly used in the wine industry in the early 1940s. However, these did not prevent spillage caused by diurnal expansion and contraction of the brine, so during warm weather it was necessary to add fresh brine to the barrels daily if the film yeasts were to be held to a minimum.
Expansion bungs were devised to minimize spillage. The first such bungs were improvised by cutting the bottoms out of 0.95 liter (1 qt) size carbonated beverage or whiskey bottles and inserting the necks m rubber stoppers of a size suitable to fit the side opening of the barrels.
This device controlled expansion and contraction of the brine without spillage. However, breakage was a problem. Finally, a plastic bung was developed. This more durable bung 15.2 cm (6 inches) in diameter, provided a constant air surface to total volume ratio of 182.5 cm2 (28.3 inches2) to 189.3 liters (50 gal.) of olives and brine
The opening in the removable top of the plastic bottle used for fermentation of Spanish-type green olives has the same diameter as that of the plastic expansion bung just described. The advantage of the plastic bottle is quite obvious—182.5 cm2 (28.3 inches2) exposed air surface to 1514 liters (400gal.) of olives and brine.
The amount of brine exposed to the air can be reduced further by floating a circular disk of slightly less diameter on the surface of the brine in the top opening. Exposure to air can be entirely eliminated by floating a 2.5 cm (1 inch) layer of paraffin, microcrystalline wax, or “vaspar,” a 1 to 1 mixture of paraffin and vaseline.
Results reported by Vaughn and Martin (1971) have shown a significant increase in acid production of olives fermented in the plastic bottles as compared with those olives fermented in 189,3 liter (50 gal.) barrels or in 757 liter (200 gal.) plastic liners held in wooden shells which also have been used in the industry.
By a combination of control measures, it is possible and also practical to complete the Spanish-type green olive fermentation in 3 to 4 weeks under ideal commercial conditions (see Fig. 6.4). To accomplish this it is necessary to use control measures which require maintenance of a brine temperature of 24% to 30°C (75° to 86°F); abstinent of the salt concentration of the acidified brine to 5% to 7% (W/V); to ensure the presence of desirable lactic acid bacteria in the brine (use of a starter, if indicated); and the addition of supplementary sugar to the brine to ensure that an acidity of at least 0.8% (total acid calculated as grams lactic acid per 100 ml brine) is produced. Maintenance of nearly anaerobic conditions is mandatory.
5. Spoilage Problems of Fermented Olives:
Bacteria, yeasts, and molds may cause spoilage of olives at any time after the harvest and the final packaging of the olives. All olives, processed black- and green-ripe olives, as well as the brined olives that undergo the lactic acid fermentation, are subject to microbial attack.
The commonest and, consequently, best-known spoilage types caused by microorganisms are gassy deterioration, malodorous fermentation and tissue softening. Of these, gassy fermentation and softening occur most frequently and under the widest variety of conditions; in all stages of the lactic fermentation as well as in olives undergoing “direct cure” for canning.
Gassy, “Floater,” or “Fish-Eye” Spoilage:
This abnormality is characterized by the development of blisters resulting from the accumulation of gases which cause separation of the skin from the flesh of the olives and by the formation of fissures or gas pockets which may extend to the pits of the fruit (Gas pockets are never found when blisters caused by too concentrated or too warm lye treatment solutions are observed so this chemical deterioration should not be confused with the biological one under consideration).
Through the extensive studies of Cruess and Guthier (1923) Alvarez (1926) Tracy (1934), and Vaughn and his students, it is well established that the Worm bacteria are chiefly responsible for blister and gas pocket formation. All of the species of coliform bacteria have been implicated in gassy spoilage except Escherichia coli.
Cultures of Bacillus polymyxa and B. macerans also cause gassy deterioration of olives. These species also may cause softening of olives. The gas-forming pseudomonad Aero-monas liquefaciens also may form gas pockets in olives but like the bacilli is equally important as a softening organism.
Saccharolytic species of the anaerobes of the genus Clostridium also cause a violent gassy fermentation in olives as shown by Gililland and Vaughn (1943) These anaerobes may also be involved in softening of olives but are more important for the malodorous spoilage they cause (butyric fermentation and zapatera)
It was formerly the contention of the author that only those bacteria which produce hydrogen were dangerous. There always are exceptions. Recently, Vaughn et al. (1972) associated yeasts of the genera Saccharomyces and Hansenula with gassy fermentation and softening in olives.
These yeasts produced typical gas blisters but did not causes fissure formation- Two of the species Saccharomyces kluyveri and S. oleaginosus, also caused severe softening of olives. The cultures of Hansenula were not pectolytic.
Control measures, for the most part, include sanitation, regulated control of the fermentation by reducing the PH by acidification, ensuring a population of desirable lactic acid bacteria, and, in the case of processed olives for canning, use of pasteurization. Control measures are discussed in more detail by Vaughn (1954).
Malodorous Fermentations:
There are 3 extremely malodorous fermentations, caused by bacteria, which develop in olives. They are the butyric acid fermentation, hydrogen sulfide fermentation, and zapatera spoilage. The butyric acid fermentation has been associated with olives since Hayne and Colby (1895) first recorded olive spoilage by the “butyric ferment.”
This abnormal fermentation is characterized by its butyric acid or rancid butter odor during the initial stages of the development of this malodorous abnormality. However, as the spoilage progresses, the odor intensifies and finally results in a very malodorous stench.
Gililland and Vaughn (1943) first isolated pure cultures of anaerobic, spore-forming butyric acid bacteria from samples of butyric spoiled olives. All of the cultures were found to be of the saccharolytic but not proteolytic types of the genus Clostridium. Most of the cultures were closely related to or identical with the species Clostridium butyricum.
Sevillano olives formerly were prone to develop butyric fermentations because the salt concentration was kept lower in order to avoid salt shrivel. However, if the salt concentration is kept in the range now used by the industry (7 to 8% NaCl W/V) the butyric fermentation cannot develop.
It always occurred during the initial stage of fermentation in storage, Sicilian- and Spanish-type olives and generally affecting Sevillano olives, but the other varieties as well, if the salt was in the range of 5% and the pH value was at 4.5 or above. At present, olives affected by the butyric fermentation are rarely found.
Hydrogen sulfide fermentation of olives is characterized by the identifying odor of H2S gas. At first the odor may be slight, but as the fermentation progresses the odor intensifies and is reminiscent of the smell of rotten eggs. Black brines may occur in this fermentation if sufficient contaminating ferrous iron is present to cause the formation of the black iron sulfide.
Black brines have been observed by the author in both storage and Sicilian-type olive brines. Chalky white brines have also been observed to occur in Spanish-type olives when contaminating zinc from galvanized pipelines, buckets, and barrel hoops got into the brine and formed the whitish zinc sulfide. The majority of hydrogen sulfide fermentation brines shows either kind of sulfide formation but is clear or has a microbial turbidity.
Early published reports on the occurrence of hydrogen sulfide fermentation in brined olives is lacking, so there is no authenticated record of the first hydrogen sulfide fermentation occurring in California olive brines. The author first observed such fermentations and recognized them as hydrogen sulfide fermentations during the 1937 harvest season in the upper Sacramento Valley. In all probability, this fermentation has occurred for as long as olives have been brined in California or elsewhere.
From 1937 on, periodic attempts were made to isolate sulfate reducing bacteria from sulfureted brines. “Mineral” autotrophic enrichments could be carried through several transfers but eventually failed. It was not until the study made by Levin and Vaughn (1966) that it was recognized that the “mineral” media used in the earlier studies lacked essential nutrients now recognized as necessary for perpetuating the growth of most, if not all, strains of sulfate-reducing vibrios.
Levin and Vaughn associated the halophilic Desulfovibrio aestuarii with the hydrogen sulfide fermentation of fermenting storage and Sicilian-type olives involving the Sevillano variety. Control of the sulfate-reducing D. aestuarii is accomplished by acidification to a pH value below 5.5 either by direct addition of acid or control of the fermentation. Salt concentration is of no control value because some of the bacteria grow in the presence of 12 to 14% NaCl (W/V).
Avoidance of hydrogen sulfide fermentation is effective if open tanks of fermenting olives are pumped over to recirculate the brine after each heavy rain to ensure an inhibitory pH in the upper layers of the tank. Sulfureted olives may be salvaged by replacing the brine and then aerating violently to oxidize the remaining hydrogen sulfide. More than one change of brine and aeration may be necessary. Once the odor is depleted then the brine must have the pH value adjusted to a safe level below 5.5.
“Zapatera,” another malodorous fermentation of olives, apparently was first described by Cruess (1924), who had observed the abnormality in Spanish green olives while on a visit to Spain. This spoilage occurs in all types of brined olives and, to the author’s knowledge, is found in all olive growing areas of the world where olives are brined.
The off-odor associated with zapatera at first is described as “cheesy” or “sagey,” but as the spoilage progresses the “cheesy” odor disappears, and as the odor further intensifies, it develops into an unmistakably characteristic foul, fecal-like stench.
Under the conditions that prevail in California, zapatera spoilage, unlike the butyric fermentation already described, occurs when the desirable lactic acid fermentation is allowed to stop before the pH of the brine has decreased to a value of 4.5 or a little less. At the start of the spoilage the pH of the infected brine increases while the titratable acidity decreases. Continuous loss in acidity is observed as the spoilage progresses.
The implication of bacteria as the cause of zapatera at first was confused. Smyth (1927), the first to investigate the problem, concluded that the spoilage was caused by one or more of a group of spore-forming, proteolytic, facultative rods normally present in the soils of Andalusia, an olive growing area in Spain.
Soriano and Soriano (1946) and Soriano (1955) claimed that zapatera spoilage in Argentina was caused by the sulfate-reducing bacterium Desulfovibrio desulfuricans. However, hydrogen sulfide odor is either not detectable or, at most, is not pronounced in the case of zapatera, either in California olives or olives from other parts of the world where olives are grown. Therefore, it was doubtful that these investigators had isolated the bacterium capable of causing the spoilage.
Because of the previous inability to isolate bacteria capable of causing zapatera, the author and his associates decided to investigate the acidic constituents of normal and zapatera fermented brines. It was found by Delmouzos et al. (1953) that normal fermented olives contained acetic, lactic, and some succinic acid, whereas zapatera brines also contained formic, propionic, butyric, valeric, isovaleric, caproic, and caprylic acids.
This information suggested the association of species of the genus Clostridium with zapatera spoilage, especially since previous investigation by Bhat and Barker (1947) and Tabachnick and Vaughn (1948) had demonstrated lactate utilization to be accentuated by the presence of acetate. Further study by Kawatomari and Vaughn (1956) associated a number of species of Clostridium with zapatera spoilage. Two saccharolytic, proteolytic species, C. bifermentans and C. sporogenes, predominated among the cultures isolated and studied.
However, the association of species of Clostridium with zapatera spoilage did not give complete explanation of the etiology of the spoilage. Propionic acid is one of the abnormal acids found in the spoiled brines. Clostridium propionicum was not found in the study made by Kawatomari and Vaughn so an additional investigation was made to determine whether species of Propionibacterium also might be involved in the spoilage.
Plastourgos and Vaughn (1957) found species of Propionibacterium including representative isolates of P. pentosaceum and P. zeae to be abundant in zapatera brines. All produce propionic acid from lactate in culture media and in olive brines. It is obvious; therefore, that the propionic acid bacteria are the cause of the cheesy odor that occurs early in zapatera spoiled olives. It is also obvious that the spoilage is caused by the participation of species of at least 2 genera of bacteria, Clostridium and Propionibacterium, and is but a manifestation of nature’s attempt to cause mineralization of the olives.
Control to prevent zapatera spoilage involves direction of the lactic fermentation until the pH value is at least at 4.0, and preferably 3.8 or below.
Zapatera spoiled olives should not be sold because once the spoilage develops, the odors cannot be removed. Nevertheless, zapatera olives still are found in commercial channels throughout the world.
6. Softening Spoilage
of Fermented Olives:
The softening of olives is usually caused by the activity of pectolytic microorganisms. Bacteria, molds, and yeasts all have been incriminated in the problem. However, the form and texture of fresh olives may be changed by use of too concentrated sodium hydroxide solutions, by frosting, or by heating. Therefore, it is not always easy to determine whether the softening is of microbial origin or whether it results from chemical or physical mistreatment of the fruit.
Softening manifests itself in 1 to 3 characteristic changes in the appearance of the olives. These changes are known in the industry by the very descriptive terms “soft stem end,” “nail head,” and “sloughing,” respectively. These types of softening have been observed in all kinds of pickled olives and all varieties are susceptible.
Soft stem end spoilage is cause by a variety of bacteria, molds, and yeasts which are known to produce pectolytic enzymes which cause softening of olives. Olives are mechanically destemmed on arrival at the processing plant or in the field at harvest when gathered from the trees.
This leaves interior tissues unprotected by stem tissue, so enzymes enter through the unprotected area and cause the splitting of the pectic materials into fragments. The pectolytic attack m turn softens the stem end area and causes a puckering of the stem end. As more tissue is attacked, the softened area expands until the entire flesh of the olive has been affected and becomes soft and, many times, quite mushy.
The bacteria known to be involved in soft stem end spoilage include spore-forming species of Bacillus and Clostridium, and the Gram-negative bacteria, including representatives of the genera Aerobacter, Aeromonas, Achromobacter, Escherichia, and Paracolobactrum.
Molds (fungi) responsible for stem end softening of olives include Aspergillus, Fusarium, Geotrichum, Paecilomyces, and Penicillium.
Yeasts involved in soft stem end spoilage of olives include oxidative, pink yeasts of the genus Rhodotorula and fermenting yeasts of the genus Saccharomyces.
The bacteria all produce pectolytic enzymes that are active at a pH range of about 6.0 to 10.0 and have an optimum pH at about 8.0 to 8.5. It is obvious, therefore, that the bacterial enzymes will not be a factor in softening unless, for some reason, the fermentation is abnormally slow. On the other hand, they are very damaging in the process of preparation of olives for black- or green-ripe canned olives during the final stage of washing prior to canning. The bacterial induced soft stem end spoilage is controlled by acidification and/or control of the lactic acid fermentation and by pasteurization in the case of the ripe olive process.
The fungal and yeast enzymes on the other hand are active at pH values in the range commonly found for storage, Sicilian- and Spanish-type olives (about 3.8) and still are active at neutrality. It is certain, therefore, that the molds and yeasts are responsible for the softening of olives in brine at pH 5.5 and below.
The bacteria generally produce two pectolytic enzymes- pectin methyl esterase, which demethylates the pectin molecule to pectic acid and either endo- or exo-polygalacturonic acid trans-eliminase, which causes degradation of the polygalacturonate to either unsaturated digalacturonic acid or unsaturated trigalacturonic acid.
The yeasts known to be pectinolytic produce polygalacturonase, which degrades pectin to saturated digalacturonic acid. One mold, Aspergillus fonsecaeus, studied by Edstrom and Phaff (1964) produces pectin transeliminase, and the major end products of pectin degradation include unsaturated and methylated galacturonates. Most of the other molds produce polygalacturonase and the major end product is monogalacturonic acid.
Since the molds are aerobic, the maintenance of anaerobic conditions will control their activity. Yeast control to prevent such softening is not so simple. The experience of the author with fermenting yeast spoilage would lead one to believe that the best control measure is to direct the lactic acid fermentation to completion. The yeast episode described by Vaughn et al. (1972) was the first encounter with fermenting, pectolytic yeasts in 35 years of experience with olives by the author.
Nail head spoilage is characterized by the formation of a concave depression under the skin of the olive which causes the skin to depress into the concave area which generally is nearly devoid of tissue. The depressions are from about 0.32 to 0.64 cm (1/8 to 1/4 inches) in diameter. The spoilage occurs infrequently, and the cause is not perfectly known.
The author has reproduced a type of nail head spoilage by bruising but not penetrating the skin of fresh olives, then fermenting them in brine and making the olives into canned black ripe olives. The resultant spoilage was identical to that observed with commercial samples. It may be that nail head spoilage is a purely physical defect due to a combination of bruising and resultant physiological deterioration of the bruised area.
Sloughing spoilage of olives occurs most frequently with olives undergoing the black-ripe process for canned olives. However, the Gram-negative pectolytic bacteria described in 1969 did not cause rupture of the skin and sloughing of the flesh, although the olives became soft and mushy.
A search was made for free-living, cellulase-producing bacteria from olives undergoing sloughing. Cellulolytic bacteria were associated with sloughing spoilage of California ripe olives by Patel and Vaughn (1973). The most active cellulolytic bacterium was identified as Cellulomonas flavigena.
Other cellulolytic bacteria studied included species of Xanthomonas, Aerobacter, Escherichia, Kurthia, Micrococcus, and Alcaligenes. All cultures produced skin rupture and sloughing of the flesh of sterile olives to a degree but the cellulomonad was the most active. Control is simple. Pasteurization may be used or the washing cycle can be reduced from 4 to not more than 3 days.
Species of Fusarium isolated from storage and Sicilian-type brines by Balatsouras and Vaughn (1958) also were found to be cellulolytic, as well as pectinolytic. It is conceivable, therefore, that they might cause some sloughing involving skin rupture, at least, which would occur in storage olives during ripe processing. Control of the fusaria would involve strict control of anaerobiosis in the fermentations.
7. Other Abnormalities
of Fermented Olives:
Two other abnormalities sometimes occur in olives. They are “yeast spot” formation in green olives and re-fermentation of bottled green olives. “Yeast spot” formation is a very common defect associated with fermented green olives in California. This abnormality is characterized by the formation of raised white spots (pimples or pustules) directly under the pores (stomata) of the olive, with a colony of microorganisms developing between the epidermis and the underlying tissues.
The Sevillano variety is prone to this abnormality. It is not confined to California but is also observed in other places where olives are fermented. “Yeast spots” also have been observed on pickled cucumbers and fermented green tomatoes. Although commonly called “yeast spots” throughout the industry, Vaughn et al. (1953) found that most, if not all, of the pustules contained lactobacilli. These bacteria were identified as Lactobacillus plantarum and L. brevis and other species closely related to the latter one. The cause of such colonization is not known, nor is any control available.
Since such olives are considered unsightly by the brokers, some loss of value of the pickled fruit is experienced, although the affected olives are perfectly normal and healthful in other respects.
Re-fermentation of glass packed olives sometimes occurs with accompanying gas formation and unsightly sedimentation caused by the growth of microorganisms. The cause of this re-fermentation is a reservoir of fermentable material still left in the olives, or the unwitting addition of fermentable sugar contained in stuffing (pimiento and almonds in particular) for pitted olives, or in various spices and other flavoring constituents added when the olives are packed in glass.
Most re-fermentation is caused by lactobacilli and, sometimes, a mixture of these bacteria and fermenting yeasts. To control refer-mentation effectively, it must be determined that all of the fermentable constituents in the olives and other added materials have been decomposed.
It is common practice to re-ferment pitted olives stuffed with pimiento or nuts for at least a month prior to finally packing the olives in glass. Pasteurization [heating to a center temperature of 85° to 88°C (185° to 190°F)] has been used. Some packers use sorbic acid (potassium sorbate) as a preservative, and vacuum seals are preferred.
Re-fermented olives are salvageable but the cost of packing and repacking is doubled because control was not practiced before the olives were packed the first time.
8. Newer Developments
in Fermentation of Olives:
Recent studies have been directed toward increasing the rate of fermentation, particularly with the Manzanilla variety. Attention has been directed toward the use of heat and studies on antibacterial substances present in olives.
Heat received the first attention. Samish et al. (1966) reported a many-fold increase in the rate of the lactic acid fermentation of green olives following a short pre-brining treatment with hot alkali which, in effect, caused lye peeling of the fruits. With this method, the rate of fermentation was stimulated when the concentration and/or the temperature of the alkali was increased.
Etchells et al. (1966) found that a pre-brining heat treatment in water, after application of lye to destroy part of the bitterness of the olives, increased the ferment-ability of the heat-shocked olives over those receiving lye treatments alone. As a result, Etchells and co-workers concluded that the ferment-ability of the different varieties depended more on their inhibitor content than any other single property they might have. Confirmatory results with such heat treatment studies have been reported by Borbolla y Alcala et al. (1971).
The notion of a microbial inhibitor in olives had been mentioned by ‘ Vaughn (1954) who speculated that oleuropein might eventually be shown to be inhibitory to the lactic acid bacteria. However, until Fleming and Etchells (1967) reported the occurrence of an inhibitor of lactic acid bacteria in green olives, there was no concrete evidence that such inhibitors might exist.
They found the inhibitor was soluble in water and ethanol, heat stable in water at 100°C, but was labile to alkaline conditions. Unfortunately, the results reported were done with frozen olives—never pickled in California because of the adverse effects on texture caused by freezing.
Shortly thereafter, Juven et al. (1968A,B) reported that the bitter gluco-side of green olives was found to inhibit the growth of lactic acid bacteria. In 1969, Fleming et al. described an inhibitor of lactic acid bacteria which was a phenolic compound devoid of acid-hydrolyzable reducing sugar but with a bitter taste.
In 1970 Juven and Henis reported the identification of the glucoside oleuropein and its aglycone as the main antibacterial components of green olives. It appears from the literature that the compounds investigated by the Israeli and United States groups were probably identical. This assumption is supported by the more detailed report by Fleming et al. (1973A) showing that two hydrolysis products of oleuropein, the aglycone and elenoic acid, inhibited the growth of lactic acid bacteria but oleuropein itself had no effect.
While the two hydrolysis products of oleuropein appear to be inhibiting to the lactic acid bacteria in vitro, there must be some reservation with regard to their inhibitory role in Spanish-type green olive fermentations. The stability of the antimicrobial factor in alkali is poor. It is common practice in California to leach lye-treated green olives with 3 or 4 changes of water in 24 hr and then add unacidified brine. When so brined the olives are quite alkaline and remain so for some time. Under such conditions it is felt that the inhibitory aglycone would be subject to further decomposition.
Also there is disagreement concerning the inhibitory activity of oleuropein. Juven et al. (1968A) reported that it inhibited the growth of different bacteria, including Lactobacillus plantarum and Leuconostoc mesenteroides. No such activity for the glucoside was recorded by Fleming et al. (1973A). Recently, Garrido-Fernandez and Vaughn (1978) made a detailed in vitro study to assess the possible utilization of oleuropein as a major source of carbon by various microorganisms associated with fermentation of olives.
It was found that both the desirable lactic acid bacteria and spoilage organisms did use oleuropein as a source of carbon, many without a significant delay in growth. It was found that increase in oleuropein from 0.2 to 0.4% (W/V) had little effect on the spoilage organisms, delaying somewhat, but not preventing, eventual growth by any of the lactic acid bacteria tested.
Another complicating factor is that the epidermis (skin or peel) of the olive is known to have a low permeability for water and gasses as shown by Duran and Tamayo (1964). The permeability of the skin is greatly increased when subjected to a hot alkali treatment, when heat shocked after normal lye treatment, or frozen. These chemical and physical methods for increasing the rate of fermentations of green olives are all subject to the same criticism—they all may adversely affect the texture of the olives so treated.
Green Olive Processing without Fermentation:
A method of salt-free storage of olives combining acidulated water (lactic and acetic acids), the food preservative sodium benzoate, and anaerobiosis was described by Vaughn et al. (1969C). This method is being used to store olives for ripe processing formerly stored in brine.
The use of salt-free storage has eliminated the problem of salt shrivel of Ascolano and Sevillano olives and improves the texture and flavor of Sevillano olives so stored. For reasons unknown the Mission variety does not store well for extended periods (6 months) but becomes softened and grainy in texture after 1 to 2 months of storage.
The method was developed to alleviate the problems of disposal of salt brines, a result of environmental protection legislation. An additional possibility exists. Vaughn and Martin (1967 -1968) observed that green olives lose much of their bitterness when stored for several months in this salt-free environment. The olives, when repacked with the proper salt brine to balance the acidity of the fruits, have very good organoleptic properties. On the basis of “blind” tasting they have passed as green fermented olives.