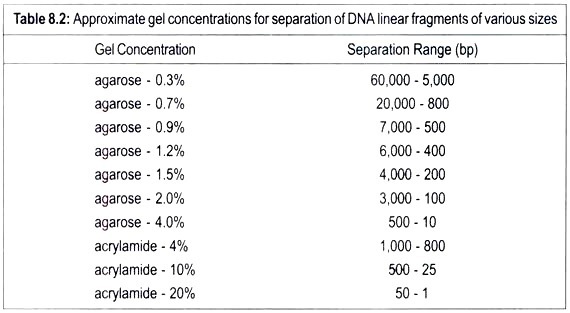
This article throws light upon the three types of recombinant vaccines. The three types are: (1) Subunit Vaccines (2) Attenuated Recombinant Vaccines and (3) Vector Recombinant Vaccines.
Contents
Recombinant Vaccines—General:
Recombinant DNA technology in recent years has become a boon to produce new generation vaccines. By this approach, some of the limitations (listed above) of traditional vaccine production could be overcome. In addition, several new strategies, involving gene manipulation are being tried to create novel recombinant vaccines.
Types of Recombinant Vaccines:
The recombinant vaccines may be broadly categorized into three groups:
1. Subunit recombinant vaccines:
These are the components of the pathogenic organisms. Subunit vaccines include proteins, peptides and DNA.
2. Attenuated recombinant vaccines:
These are the genetically modified pathogenic organisms (bacteria or viruses) that are made non-pathogenic and used as vaccines.
3. Vector recombinant vaccines:
These are the genetically modified viral vectors that can be used as vaccines against certain pathogens. Some of the developments made in the production of recombinant vaccines against certain diseases are briefly described.
Type # 1. Subunit Vaccines:
As already stated, subunit recombinant vaccines are the components (proteins, peptides, DNAs) of the pathogenic organisms. The advantages of these vaccines include their purity in preparation, stability and safe use. The disadvantages are — high cost factor and possible alteration in native conformation. Scientists carefully evaluate the pros and cons of subunit vaccines for each disease, and proceed on the considered merits.
Hepatitis B:
Hepatitis B is a widespread disease in man. It primarily affects liver causing chronic hepatitis, cirrhosis and liver cancer. Hepatitis B virus is a 42 nm particle, called Dane particle. It consists of a core containing a viral genome (DNA) surrounded by a phospholipid envelope carrying surface antigens (Fig. 16.1 A).
Infection with hepatitis B virus produced Dane particles and 22 nm sized particles. The latter contain surface antigens which are more immunogenic. It is however, very difficult to grow hepatitis B virus in mammalian cell culture and produce surface antigens.  The gene encoding for hepatitis B surface antigen (HBsAg) has been identified. Recombinant hepatitis B vaccine as a subunit vaccine, is produced by cloning HbsAg gene in yeast cells. Saccharomyces cerevisiae, a harmless baking and brewing yeast, is used for this purpose (Fig. 16.1B). The gene for HBsAg is inserted (pMA 56) which is linked to the alcohol dehydrogenase promoter. These plasmids are then transferred and cultured.
The gene encoding for hepatitis B surface antigen (HBsAg) has been identified. Recombinant hepatitis B vaccine as a subunit vaccine, is produced by cloning HbsAg gene in yeast cells. Saccharomyces cerevisiae, a harmless baking and brewing yeast, is used for this purpose (Fig. 16.1B). The gene for HBsAg is inserted (pMA 56) which is linked to the alcohol dehydrogenase promoter. These plasmids are then transferred and cultured.
The cells grown in tryptophan, free medium are selected and cloned. The yeast cells are cultured. The HBsAg gene is expressed to produce 2nm sized particles similar to those found in patients infected with hepatitis B. (These particles are immunoreactive with anti-HBsAg antibodies). The subunit HBsAg as 22 nm particles can be isolated and used to immunize individuals against hepatitis B.
Hepatitis B vaccine-the first synthetic vaccine:
In 1987, the recombinant vaccine for hepatitis B (i.e. HBsAg) became the first synthetic vaccine for public use. It was marketed by trade names Recombivax and Engerix-B. Hepatitis B vaccine is safe to use, very effective and produces no allergic reactions. For these reasons, this recombinant vaccine has been in use since 1987.
The individuals must be administered three doses over a period of six months. Immunization against hepatitis B is strongly recommended to anyone coming in contact with blood or body secretions. All the health professionals—physicians, surgeons, medical laboratory technicians, nurses, dentists, besides police officers, firefighters etc., must get vaccinated against hepatitis B.
Hepatitis B vaccine in India:
India is the fourth country (after USA, France and Belgium) in the world to develop an indigenous hepatitis B vaccine. It was launched in 1997, and is now being used.
Hepatitis B vaccine tomato?
Biotechnologists have been successful in inserting hepatitis B gene into the cells of the tomato plant. These genetically engineered plants produce hepatitis B antigens. The day may not be far off to get immunized against hepatitis B by having a tomato with lunch!
Foot and Mouth Disease:
Foot and mouth disease (FMD) is a highly contagious disease affecting cattle and pigs. A formalin killed foot and mouth disease virus (FMDV) was previously used to vaccinate against this disease. The genome of FMDV is composed of a single— stranded RNA, covered by four viral proteins (VP1, VP2, VP3 and VP4). Among these, VPI is immunogenic. The nucleotide sequence encoding VPI was identified in the FMDV genome. A double- stranded complementary DNA (cDNA) from the single-stranded viral RNA (genome) was synthesized.
This cDNA was then digested with restriction enzymes and the fragments were cloned by using plasmid pBR322 in E. coli. The recombinant vaccine for FMDV in the form of viral protein 1 was used to vaccinate animals. However, VPI vaccination was found to be less effective than that of the whole virus in protecting FMD. Further, studies are being pursued to improve the efficiency of subunit vaccine.
The concept of peptide vaccines:
Theoretically, it is expected that only small portions of a given protein (i.e., domains) are immunogenic and bind to antibodies. Logically, it is possible to use short peptides that are immunogenic as vaccines. These are referred to as peptide vaccines.
Peptide vaccines for foot and mouth disease:
Some details on the FMD are described above. The domains of viral protein I (VPI) of FMDV were chemically synthesized. From the C-terminal end of VPI, amino acids 141 to 160, 151 to 160 and 200 to 213, and from N-terminal end, amino acids 9 to 24, 17 to 32 and 25 to 41 were synthesized.
Each one of these short peptides (domains) was bound to the surface of a carrier protein (Fig. 16.2) and used as a vaccine. Among the peptides used, the one corresponding to amino acids 141 to 160 was found to be effective in immunizing guinea pigs against FMD. In addition, when two peptides were joined together (amino acids 141 to 158, and amino acids 200 to 213), they served as more efficient recombinant vaccines.
The success so far to use recombinant peptides as vaccines has been very limited. This is mainly because a short peptide usually is not enough to be sufficiently immunogenic, since it may not have the same conformation as that of the original viral particle. However, scientists continue their search for specific, inexpensive and safe synthetic peptide vaccines for various diseases.
Herpes Simplex Virus:
Herpes simplex virus (HSV) is an oncogenic (cancer-causing) virus. In addition, it also causes sexually transmitted diseases, encephalitis and severe eye infections. Attempts have been made to produce subunit vaccines against HSV. An envelope glycoprotein D (gD) of HSV that can elicit antibody production has been identified. This is a membrane bound protein, and difficult to isolate and purify. The glycoprotein D was modified by deleting the trans membrane portion of the protein (Fig. 16.3) and the gene was modified.
This gene for gD was cloned in a mammalian vector and expressed in Chinese hamster ovary (CHO). The advantage here is that the protein can get glycosylated (unlike in E. coli system). In the experimental trials, the modified form of gD was found to be effective against HSV.
Tuberculosis:
Tuberculosis is caused by the bacterium Mycobacterium tuberculosis. It is often fatal, and as per some estimates nearly 3 million deaths occur every year due to this highly infectious disease. Antibiotics are used to treat tuberculosis. However, drug-resistant M. tuberculosis strains have been developed making the drug therapy sometimes ineffective. Vaccination for tuberculosis is therefore, advocated.
Bacillus Calmette-Guerin (BCG) vaccine:
In some countries, particularly the developing ones, BCG vaccine is widely used to protect against tuberculosis. However, countries like United States have not approved BCG vaccination for various reasons. BCG vaccine itself causes tuberculosis in some individuals (AIDS victims) and the vaccinated people respond positively for laboratory diagnosis of tuberculosis.
Subunit vaccines:
The secretory (extracellular proteins of M. tuberculosis have been purified and used for immuno-protection against tuberculosis. Of about 100 such proteins, six were found to be useful (either individually or in combination) to immunize guinea pigs. Attempts are underway to develop recombinant subunit vaccine against tuberculosis.
Meningitis:
Group B strain of meningococci, namely Neisseria meningitidis causes meningitis in adolescents and young adults. Meningitis is characterized by inflammation of the membranes covering brain and spinal cord. The symptoms include headache, photophobia, irritability, and neck stiffness.
Pizza et al (2000) made a novel approach to develop a vaccine against meningitis. They identified 350 proteins (potential protective antigens) and the entire sequence of genome coding for these proteins in N. meningitidis. All the 350 candidate antigens were expressed in E. coli, purified and used to immunize mice. A good bactericidal antibody response was observed in these mice.
AIDS (Acquired Immunodeficiency Syndrome):
AIDS is a retroviral disease caused by human immunodeficiency virus (HIV). This disease is characterized by immunosuppression, neo-plasma and neurological manifestations. AIDS is invariably fatal, since as of now there is no cure. Development of a vaccine against AIDS is a top priority by DNA technologists world over. In fact, vaccines are being continuously developed and field tested, although there has been no success so far.
Subunit vaccines:
The development of two subunit vaccines, specifically the glycoproteins of HIV envelope is described here. The functions of gp120 and gp41 of HIV are illustrated in Fig. 16.4A. The glycoprotein gp120 projects out of the HIV envelope while the other glycoprotein gp41 lies beneath gp120.
On entering the body, the HIV binds to the host cells (T-lymphocytes) by attaching gp120 to the CD4 receptor sites on the cell surface. This attachment uncovers gp41 molecules and the viral envelope. Now gp41 binds to the host cell surface and opens a passage for the entry of the virus into the cell.
Biotechnologists have isolated the genes for gp120 and gp41 and inserted them into the bacterium E. coli. These bacterial cells produce gp120 and gp41 that can be used as recombinant vaccines against AIDS. The action of gp120 and gp41 in immunizing host T-lymphocytes is depicted in Fig. 16.4B.
The gp120 molecules stimulate the host immune system to produce anti- gp120 antibodies. These antibodies bind to gp120 and prevent its attachment to CD4. In a comparable manner, gp41 molecules also result in the production of anti-gp41 antibodies. These antibodies also bind to gp41 and block the virus- host cell union. The net result of using gp120 and gp41 vaccines is that the entry of HIV into the host cells is prevented.
Vaccine against AIDS— not yet a reality:
The description of vaccine development against AIDS (given above), which appears attractive is not so simple. The most important limitations are that the HIV has high frequency of mutations. Therefore the vaccines developed cannot bind to the new virus (i.e., mutated one). In addition, gp120 and gp41 are very poor stimulators of immune system. Despite these limitations, scientists have not lost hope, and continue their research to develop vaccines against AIDS.
DNA Vaccines (Genetic Immunization):
Genetic immunization by using DNA vaccines is a novel approach that came into being in 1990. The immune response of the body is stimulated by a DNA molecule. A DNA vaccine consists of a gene encoding an antigenic protein, inserted onto a plasmid, and then incorporated into the cells in a target animal.
The plasmid carrying DNA vaccine normally contains a promoter site, cloning site for the DNA vaccine gene, origin of replication, a selectable marker sequence (e.g. a gene for ampicillin resistance) and a terminator sequence (a poly—A tail).
DNA vaccine—plasmids can be administered to the animals by one of the following delivery methods.
i. Nasal spray
ii. Intramuscular injection
iii. Intravenous injection
iv. Intradermal injection
v. Gene gun or biolistic delivery (involves pressure delivery of DNA-coated gold beads).
DNA Vaccine and Immunity:
An illustration of a DNA vaccine and the mechanism of its action in developing immunity is given in Fig. 16.5. The plasmid vaccine carrying the DNA (gene) for antigenic protein enters the nucleus of the inoculated target cell of the host. This DNA produces RNA, and in turn the specific antigenic protein. The antigen can act directly for developing humoral immunity or as fragments in association with major histocompatibility class (MHC) molecules for developing cellular immunity.
Humoral immunity:
As the antigen molecules bind to B- lymphocytes, they trigger the production of antibodies which can destroy the pathogens. Some of the B-lymphocytes become memory cells that can protect the host against future infections.
Cellular immunity:
The protein fragments of the antigen bound to MHC molecules can activate the cytotoxic T-lymphocytes. They are capable of destroying the infected pathogenic cells. Some of the activated T-lymphocytes become memory cells which can kill the future infecting pathogens.
Complementary DNA vaccines:
For genetic immunization, complementary DNA (cDNA) vaccines can also be used. Some workers have successfully used cDNA as vaccines e.g. immunization of mice against influenza.
DNA vaccines for production of antigens and antibodies:
A novel approach for the production of antigens as well as antibodies by DNA vaccine was developed in 1997. In the experiments conducted in mice, researchers injected plasmids containing the genes for malarial parasite and also the genes for the antibodies against malarial parasite. The B-lymphocytes 4 of these mice performed a double duty, and produced antigens for and antibodies against malarial parasite.
The antigens stimulate to produce more and more antibodies. The antibodies so produced react with malarial parasite. The generation of antigens and antibodies by using a DNA vaccine is a recent development in immunology, and is referred to as antigenic antibody approach of DNA vaccine.
Screening of pathogenic genome for selecting DNA vaccines:
The ultimate goal of scientists is to choose the right DNA fragment from the pathogen to serve as a vaccine for the strongest immune response against the invading pathogen. For this purpose, the pathogen’s DNA can be broken into fragments and a large number of vaccines DNA—plasmids can be prepared.
The immune response for each one of the DNA vaccines can be studied by injecting the pathogen. By screening the DNA fragments of the pathogenic genome, it is possible to choose one or few DNA vaccines that can offer maximal immune protection.
Advantages of DNA vaccines:
There are several advantages of using DNA vaccines in immunization:
1. The tedious and costly procedures of purifying antigens or creating recombinant vaccines are not necessary.
2. DNA vaccines are very specific in producing the target proteins (antigens or antibodies). Thus, they trigger immune response only against the specific pathogen.
3. In general, DNA vaccines elicit much higher immune response compared to other kinds of vaccines.
4. DNA vaccines are more stable for temperature variations (low or high) than the conventional vaccines. Thus, the storage and transport problems associated with vaccines are minimal.
5. The delivery methods to the host are simpler for DNA vaccines.
Disadvantages of DNA vaccines:
1. The fate of the DNA vaccine in the host cells is not yet clear. There is a possibility of this DNA getting integrated into the host genome and this may interrupt the normal functions.
2. There also exists a danger of cancer due to DNA vaccines.
3. The post-translational modification of the gene (DNA vaccine) product in host cells may not be the same as that found in the native antigen.
Present status of DNA vaccines:
Since 1990, several groups of workers world-over have been trying to develop DNA vaccines against various diseases in experimental animals. Genetic immunization has been done against a number of pathogenic organisms. These include influenza A virus, rabies virus, hepatitis B virus, bovine herpes virus, HIV type I, and Plasmodium species (malarial parasite). It must be noted that DNA vaccines have not been tried in humans for obvious reasons. The most important being the unknown risks of these foreign DNAs in human subjects.
RNA Vaccines:
Several workers are trying to use RNA molecules as vaccines. These RNAs can readily synthesize the antigenic proteins and offer immunity. But unfortunately, RNAs are less stable than DNAs. This poses a big problem for RNA vaccine manufacture and distribution. Therefore, the progress in the development of RNA vaccines has been rather slow compared to DNA vaccines.
Plants as Edible Subunit Vaccines:
Plants serve as a cheap and safe production systems for subunit vaccines. The edible vaccines can be easily ingested by eating plants. This eliminates the processing and purification procedures that are otherwise needed. Transgenic plants (tomato, potato) have been developed for expressing antigens derived from animal viruses (rabies virus, herpes virus). A selected list of recombinant vaccines against animal viruses produced in plants is given in Table 16.2.
Edible vaccine production and use:
The production of vaccine potatoes is illustrated in Fig. 16.6. The bacterium, Agrobacterium tumefaciens is commonly used to deliver the DNA (genetic material) for bacterial or viral antigens. A plasmid carrying the antigen gene and an antibiotic resistance gene are incorporated into the bacterial cells (A. tumefaciens).
The cut pieces of potato leaves are exposed to an antibiotic which can kill the cells that lack the new genes. The surviving cells (i.e., gene altered ones) can multiply and form a callus (clump of cells). This callus is allowed to sprout shoots and roots, which are grown in soil to form plants.
In about three weeks, the plants bear potatoes with antigen vaccines. The first clinical trials in humans, using a plant- derived vaccine were conducted in 1997. This involved the ingestion of transgenic potatoes with a toxin of E. coli causing diarrhea.
Type # 2. Attenuated Recombinant Vaccines:
In the early years of vaccine research, attenuated strains of some pathogenic organisms were prepared by prolonged cultivation — weeks, months or even years. Although the reasons are not known, the infectious organism would lose its ability to cause disease but retains its capability to act as an immunizing agent. This type of approach is almost outdated now.
It is now possible to genetically engineer the organisms (bacteria or viruses) and use them as live vaccines, and such vaccines are referred to as attenuated recombinant vaccines. The genetic manipulations for the production of these vaccines are broadly of two types:
1. Deletion or modification of virulence genes of pathogenic organisms.
2. Genetic manipulation of non-pathogenic organisms to carry and express antigen determinants from pathogenic organisms.
The advantage with attenuated vaccines is that the native conformation of the immunogenic determinants is preserved; hence the immune response is substantially high. This is in contrast to purified antigens which often elicit poor immunological response.
Some of the important attenuated vaccines developed by genetic manipulations are briefly described.
Cholera:
Cholera is an intestinal disease characterized by diarrhea, dehydration, abdominal pain and fever. It is caused by the bacterium, Vibrio cholera. This pathogenic organism is transmitted by drinking water contaminated with fecal matter. Cholera epidemics are frequently seen in developing countries where the water purification and sewage disposal systems are not well developed.
On entering the small intestine, V. cholera colonizes and starts producing large amounts of a toxic protein, a hexameric enterotoxin. This enterotoxin stimulates the cells lining intestinal walls to release sodium, bicarbonate and other ions. Water accompanies these ions leading to severe diarrhea, dehydration, and even death.
The currently used cholera vaccine is composed of phenol-killed V. cholera. The immuno-protection, lasting for 3-6 months is just moderate. Attempts are being made to develop better vaccines. The DNA technologists have identified the gene encoding enterotoxin (toxic protein). Enterotoxin, an hexamer, consists of one A subunit and five identical B subunits. The A subunit has two functional domains-the A1 peptide which possesses the toxic activity and A2 peptide that joins A subunit to B subunits.
By genetic engineering, it was possible to delete the DNA sequence encoding A1 peptide and create a new strain of V. cholera. This strain is non-pathogenic, since it cannot produce enterotoxin. The genetically engineered V. cholera is a good candidate to serve as an attenuated vaccine.
Creating a new strain of V. cholera:
The development of a new strain of Vibrio cholera that can effectively serve as an attenuated recombinant vaccine is depicted in Fig. 16.7, and briefly described below.
1. A tetracycline resistance gene was inserted into the A1 peptide sequence of V. cholera chromosome. This destroys the DNA sequence encoding for A1 peptide, besides making the strain resistant to tetracycline. Unfortunately, the tetracycline resistant gene is easily lost and the enterotoxin activity is restored. Because of this, the new strain of V. cholera as such cannot be used as a vaccine.
2. The DNA sequence of A1 peptide is incorporated into a plasmid, cloned and digested with restriction enzymes (Clal and Xbal). In this manner, the A1 peptide coding sequence is deleted (the DNA encoding for 183 of the 194 amino acids of the A1 peptide is actually removed). By using T4 DNA ligase, the plasmid is re-circularized. This plasmid contains a small portion of A1 peptide coding sequence.
3. The plasmid, containing the deleted A1 peptide sequence is transferred by conjugation into the V. cholera strain carrying a tetracycline resistance gene.
4. Recombination can occur between the plasmid (containing a small portion of peptide A1 coding sequence) and the chromosome of V. cholera (carrying tetracycline resistance gene). The result of this double crossover is the formation of V. cholera containing a chromosomal DNA lacking A1 peptide DNA sequence. As the bacterium, V. cholera multiplies, the plasmids are lost in the next few generations.
5. The V. cholera cells defective in A1 peptide are selected, based on tetracycline sensitivity. It may be noted that this new strain lacks tetracycline resistance gene.
The genetically engineered V. cholera cells with deleted A1 peptide DNA sequence are quite stable. They cannot produce active enterotoxin but possess all other biochemical functions of the pathogen. This new strain of V. cholera is undergoing trials for its efficiency as a vaccine. Preliminary results indicate that this attenuated vaccine can protect about 90% of the volunteers against cholera. But there are some side effects. Scientists continue their work to develop a better vaccine against cholera.
Potato as a vehicle for cholera vaccine:
A group workers have developed a gene altered potato containing attenuated cholera vaccine. These potatoes when fed to mice induced immunity against cholera.
Salmonella Species:
The different strains of Salmonella genus are responsible for causing typhoid, enteric fever, food poisoning and infant death. Immunoprotection against Salmonella pathogens is really required. Some workers have been successful in deleting aro genes and pur genes in Salmonella.
Aro genes encode for the enzymes responsible for the biosynthesis of aromatic compounds, while pur genes encode for enzymes of purine metabolism. The new strains of Salmonella can be grown in vitro on a complete medium.
The doubly deleted strains have a very restricted growth in vivo, while they can stimulate immunological response. The genetically engineered attenuated vaccines of Salmonella have been shown to be effective as oral vaccines in experimental animals (mice, cattle, sheep, and chickens). Some workers claim that the new strain of Salmonella offers immunoprotection in humans also.
Leishmania Species:
Leishmania species are flagellated protozoan parasites and are responsible for the disease leishmaniasis. This disease is characterized by cutaneous, visceral and mucosal leisons. Leishmaniasis is transmitted by sand flies.
An attenuated strain of leishmania has been created and successfully used in mice to offer immunoprotection against leishmaniasis. In Leishmania major, the genes encoding dihydrofolate reductase-thymidylate synthase can be replaced by the genes encoding resistance to antibiotics G-418 and hygromycin.
This new strain of L. major invariably requires thymidine in the medium for its growth and multiplication. The attenuated strain of L. major can survive only a few days when administered to mice. This short period is enough to induce immunity in mice against the lesions of leishmania. However, more experiments on animals have to be carried out before the leishmania attenuated vaccine goes for human trials.
Type # 3. Vector Recombinant Vaccines:
Some of vectors can be genetically modified and employed as vaccines against pathogens.
Vaccines against Viruses- Vaccinia Virus:
Vaccinia viruses is basically the vaccine that was originally used by Jenner for the eradication of smallpox. The molecular biology of this virus has been clearly worked out. Vaccinia virus contains a double-stranded DNA (187 kb) that encodes about 200 different proteins. The genome of this virus can accommodate stretches of foreign DNA which can be expressed along with the viral genes.
The vaccinia virus can replicate in the host cell cytoplasm (of the infected cells) rather than the nucleus. This is possible since the vaccinia virus possesses the machinery for DNA replication, transcription-DNA polymerase, RNA polymerase etc. The foreign genes inserted into the vaccinia virus can also be expressed along with the viral genome. Thus, the foreign DNA is under the control of the virus, and is expressed independently from the host cell genome.
The vaccinia viruses are generally harmless, relatively easy to cultivate and stable for years after lyophilization (freeze-drying). All these features make the vaccinia virus strong candidates for vector vaccine. The cloned foreign genes (from a pathogenic organism) can be inserted into vaccinia virus genome for encoding antigens which in turn produces antibodies against the specific disease- causing agent.
The advantage with vector vaccine is that it stimulates B-lymphocytes (to produce antibodies) and T-lymphocytes (to kill virus infected cells). This is in contrast to a subunit vaccine which can stimulate only B-lymphocytes. Thus, vaccinia virus can provide a high level of immunoprotection against pathogenic organisms. Another advantage of vaccinia virus is the possibility of vaccinating individuals against different diseases simultaneously. This can be done by a recombinant vaccinia viruses which carries genes encoding different antigens.
Antigen genes for certain diseases have been successfully incorporated into vaccinia virus genome and expressed. Thus, vector vaccines have been developed against hepatitis, influenza, herpes simplex virus, rabies, angular stomatitis virus and malaria. However, none of these vaccines has been licensed for human use due to fear of safety. It is argued that recombinant vaccinia virus might create life threatening complications in humans.
Production of recombinant vaccinia viruses:
The development of recombinant vaccinia virus is carried out by a two-step procedure (Fig. 16.8).
1. Assembly of plasmid insertion vector:
Fresh vaccinia (cow pox) viruses are processed to release their DNAs. Now genes from hepatitis B virus, herpes simplex virus and influenza virus are added one after another and inserted into vaccinia virus genome. These DNA clusters are cloned in E. coli for increasing their number and to produce plasmid insertion vectors. The plasmid contains the foreign subunit genes, the natural vaccinia genes, including the promoter genes. The recombinant plasmids are isolated and purified and serve as plasmid insertion vectors.
2. Production of recombinant vaccinia viruses:
The animal cells are infected with plasmid insertion vectors and normal vaccinia viruses. As the viral replication occurs, the plasmids are taken up to produce recombinant vaccinia viruses. The plasmid insertion vector incorporates its genes into vaccinia virus genome at a place that encodes for the enzyme thymidine kinase (TK).
Thus the recombinant viruses have lost their ability to produce TK. There are two advantages of loss of TK gene. One is that it is easy to select recombined vaccinia viruses that lack TK gene and the second is that these viruses are less infectious than the normal viruses. The recombinant vaccinia viruses, released from the cultured animal cells, can be successfully used as vaccines. These live viral vaccines have some advantages over the killed or subunit vaccines.
Advantages:
1. Authenticated antigens that closely resemble natural antigens can be produced.
2. The virus can replicate in the host cells. This enables the amplification of the antigens for their action on B-lymphocytes and T-lymphocytes.
3. There is a possibility of vaccinating several diseases with one recombinant vaccinia virus.
Disadvantages:
1. The most important limitation is the yet unknown risks of using these vaccines in humans.
2. There may be serious complications of using recombinant viral vaccines in immunosuppressed individuals such as AIDS patients.
Other viral recombinant vaccines:
Most of the work on the development of live viral vaccines has been carried out on vaccinia virus. Other viruses such as adenovirus, poliovirus and varicella-zoster virus are also being tried as recombinant vaccines. Scientists are attracted to develop a recombinant poliovirus as it can be orally administered. It might take many more years for the recombinant viral vaccines to become a reality for human use.
Delivery of Antigens by Bacteria:
It is known that the antigens located on the surface of a bacterial cell are more immunogenic than the antigens in the cytoplasm. Based on this observation, scientists have developed strategies to coat the surfaces of non-pathogenic organisms with antigens of pathogenic bacteria.
Flagellin is a protein present in the fragella (thread like filaments) of Salmonella. A synthetic oligonucleotide encoding the epitope of cholera toxin B subunit was inserted into Salmonella flagellin gene. This epitope was in fact found on the flagellum surface. These flagella-engineered bacteria, when administered to mice, raised antibodies against the cholera toxin B subunit peptide. It may be possible in future to incorporate multiple epitopes (2 or 3) into the flagellin gene to create multivalent bacterial vaccines.