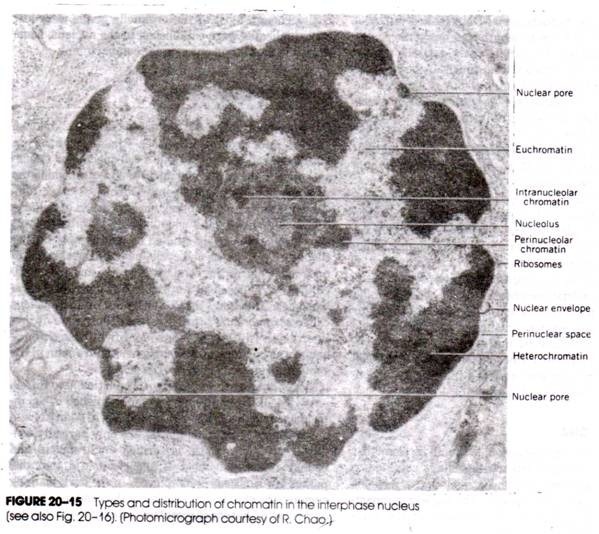
Bio-activation may be defined as a process whereby certain chemically stable compounds can be converted to chemically reactive metabolites.
For example, Parathion, an organ phosphorus pesticide, is bio-activated in the liver to Paraoxon, which is a more potent inhibitor of cholinesterase (ChE). The reactive metabolites such as Epoxides can form covalent binding to cellular proteins and cause necrosis and cancer (Fig. 8.3).
Other metabolites, such as free radicals, can cause lipid peroxidation resulting in tissue damage. For example carbon tetrachloride forms trichloromethyl radical, which causes peroxidation of polyunsatxu-ated lipid as well as covalently binds to unsturated fat and protein. The herbicide Paraquat is known to produce superoxide radicals (Halliwell et at, 1992).
Gillette and Mitchell (1975) stated that bromobenzene epoxide from the parent chemical (bromobenzene) may covalently bind to cell macromolecules but such binding may be minimal when the other metabolic routes are predominant (Fig. 8.4). The most important of these routes is the conjugation with Glutathione. This reaction is a protective mechanism against liver necrosis. Bioactivation takes place mainly in the liver and the resulting reactive metabolites induce toxicity through covalent binding with liver proteins, resulting in necrosis or cancer formation.